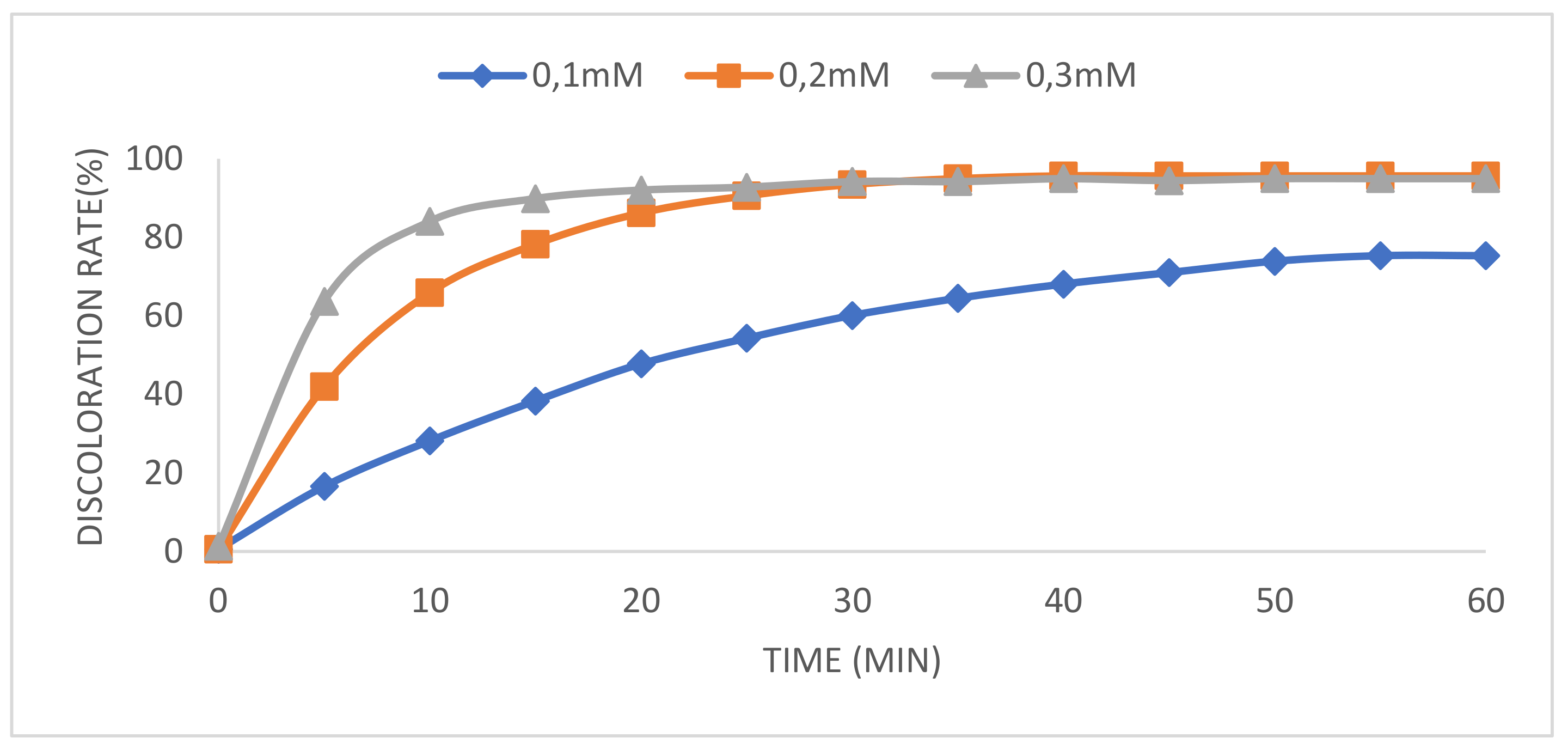
Catalysts | Free Full-Text | Decolorization and Degradation of Methyl Orange Azo Dye in Aqueous Solution by the Electro Fenton Process: Application of Optimization

Frontiers | Sorption of Perfluorooctane Sulfonic Acid Including Its Isomers to Soils: Effects of pH, Natural Organic Matter and Na2SO4

Influence of sulfate ion concentration and pH on the corrosion of Mg-Al-Zn-Mn (GA9) magnesium alloy - ScienceDirect

Charge storage mechanisms of birnessite-type MnO2 nanosheets in Na2SO4 electrolytes with different pH values: In situ electrochemical X-ray absorption spectroscopy investigation - ScienceDirect

Sodium Sulphate Anhydrous 99% pH 6-8 for Industry - China Sodium Sulfate Speckles and Sulfate Sodium Anhydrous

Nơi nhập khẩu ( bán ) Na2SO4 – Muối Sunfat PH 6-8 Trung Quốc | Công ty phân phối và cung cấp hóa chất tại TPHCM
![PDF] Nanostructured palladium hydride electrodes: from the potentiometric mode in SECM to the measure of local pH during carbonation | Semantic Scholar PDF] Nanostructured palladium hydride electrodes: from the potentiometric mode in SECM to the measure of local pH during carbonation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/fd42dbe9175be50c0653072da78e328c9c2f7899/121-Figure4.11-1.png)
PDF] Nanostructured palladium hydride electrodes: from the potentiometric mode in SECM to the measure of local pH during carbonation | Semantic Scholar
![PDF] The Influence of Solution pH on the Kinetics of Resorcinol Electrooxidation (Degradation) on Polycrystalline Platinum | Semantic Scholar PDF] The Influence of Solution pH on the Kinetics of Resorcinol Electrooxidation (Degradation) on Polycrystalline Platinum | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ece9f2a98c33aef09bd4f8cba97d64814c76eded/3-Table1-1.png)
PDF] The Influence of Solution pH on the Kinetics of Resorcinol Electrooxidation (Degradation) on Polycrystalline Platinum | Semantic Scholar

High Quality Concrete Admixture Na2so4 3% 5% 10% 18% Fdn/Sns/Snf - China Sodium Naphthalene Formaldehyde Sulfonate and Concrete Admixture

Effect of pH for the adsorption of sulfates from Na 2 SO 4 and SLS by... | Download Scientific Diagram

Na2so4 Sodium Sulphate Anhydrous 99%min Ph:6-8 - Buy Sodium Sulphate,Na2so4 Sodium Sulphate,99% Sodium Sulphate Anhydrous Prices Product on Alibaba.com

The volume of 0.1 M Na(2)SO(4) needed to be added to 10 mL of 0.1 M HCI to change the pH from 1.00 to 1.50 is : (K(2) =1.26 xx10^(-2) " for " H(2)SO(4))

Effect of Sodium Sulfate Activator on Compressive Strength and Hydration of Fly-Ash Cement Pastes | Journal of Materials in Civil Engineering | Vol 32, No 6