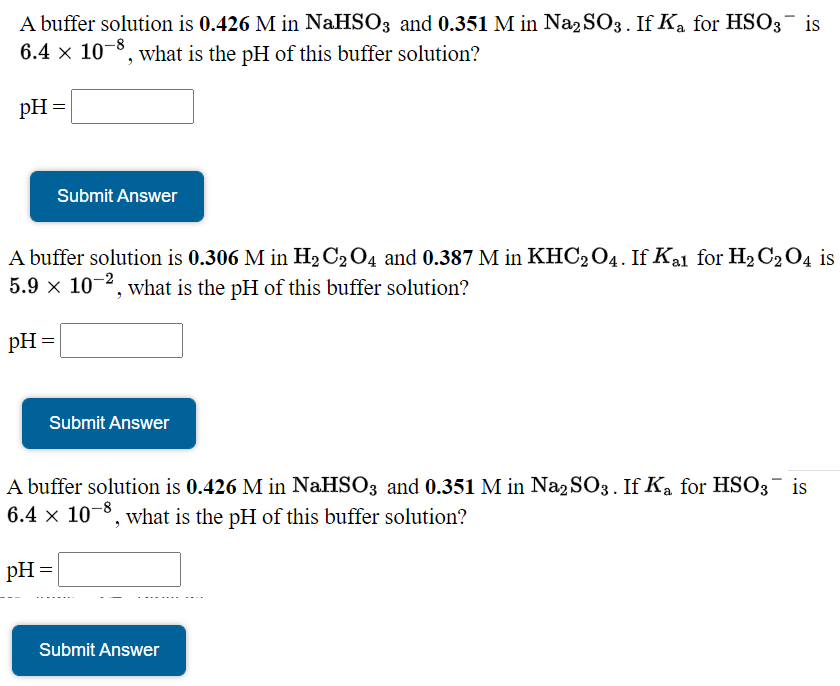
SOLVED: A buffer solution contains 0.409 M KHSO3 and 0.201 M Na2SO3. Determine the pH change when 0.120 mol KOH is added to 1.00 L of the buffer. pH after addition −

Voltammograms obtained for solutions of x M Na2SO3 (pH 12.1); x = 0.01... | Download Scientific Diagram

Effect of Na 2 SO 3 concentration and temperature on the current of the... | Download Scientific Diagram

Phase Equilibrium of the Ternary System Na2SO3–Na2SO4–H2O at 293.15, 313.15, and 353.15 K | Journal of Chemical & Engineering Data
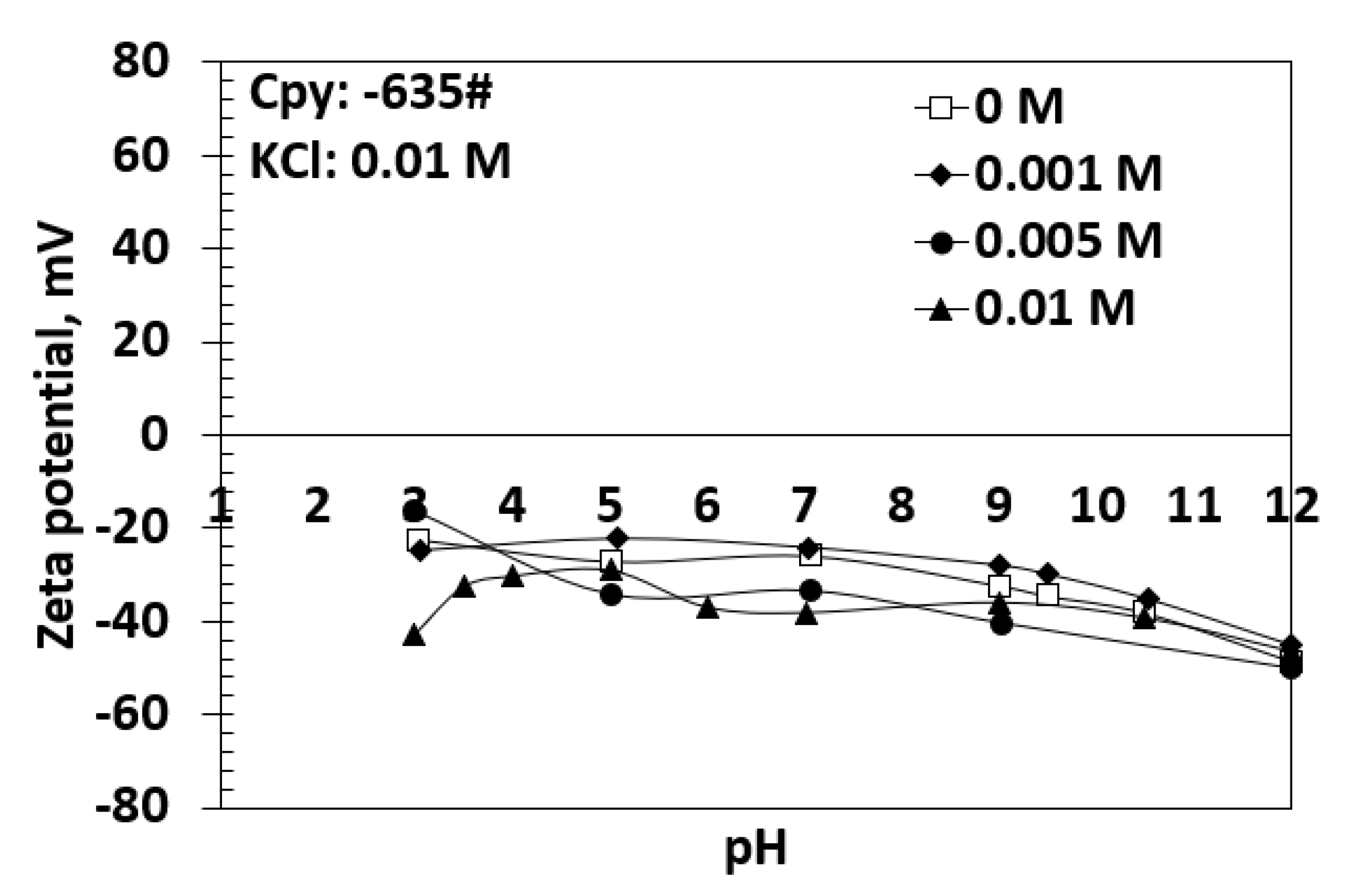
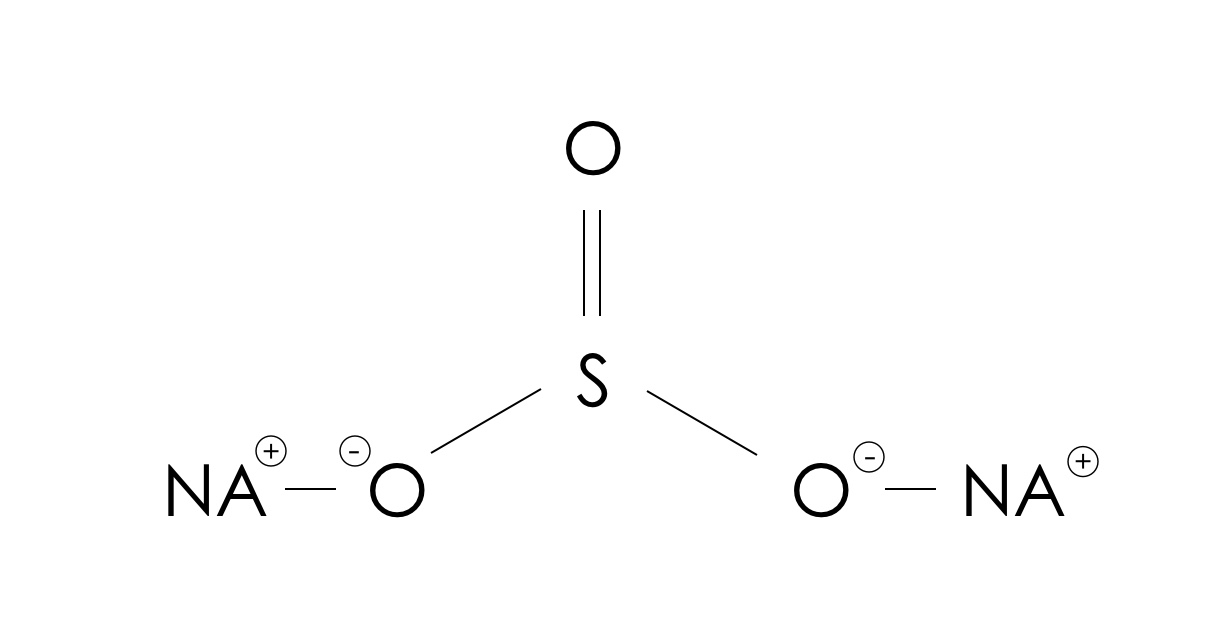
Minerals | Free Full-Text | Effects of Potassium Propyl Xanthate Collector and Sodium Sulfite Depressant on the Floatability of Chalcopyrite in Seawater and KCl Solutions

Simultaneous wet absorption of SO2 and NOX with mixed Na2SO3 and (NH4)2SO3: Effects of mass concentration ratio and pH - ScienceDirect
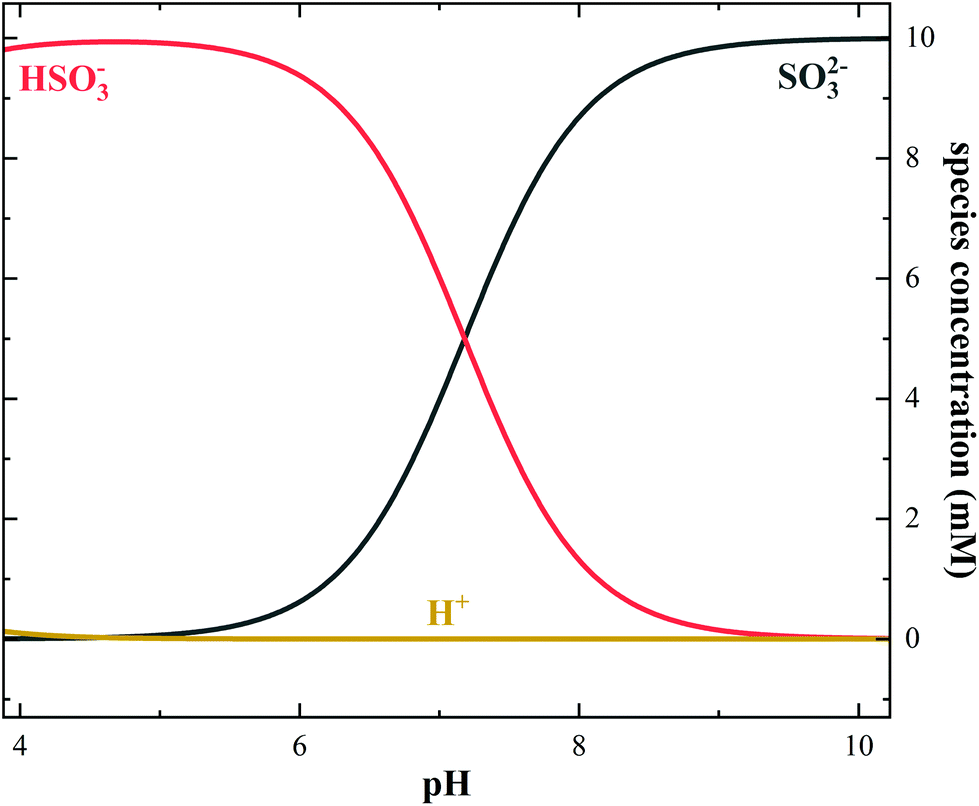
Nanosecond transient absorption studies of the pH-dependent hydrated electron quenching by HSO 3 − - Photochemical & Photobiological Sciences (RSC Publishing) DOI:10.1039/C9PP00063A
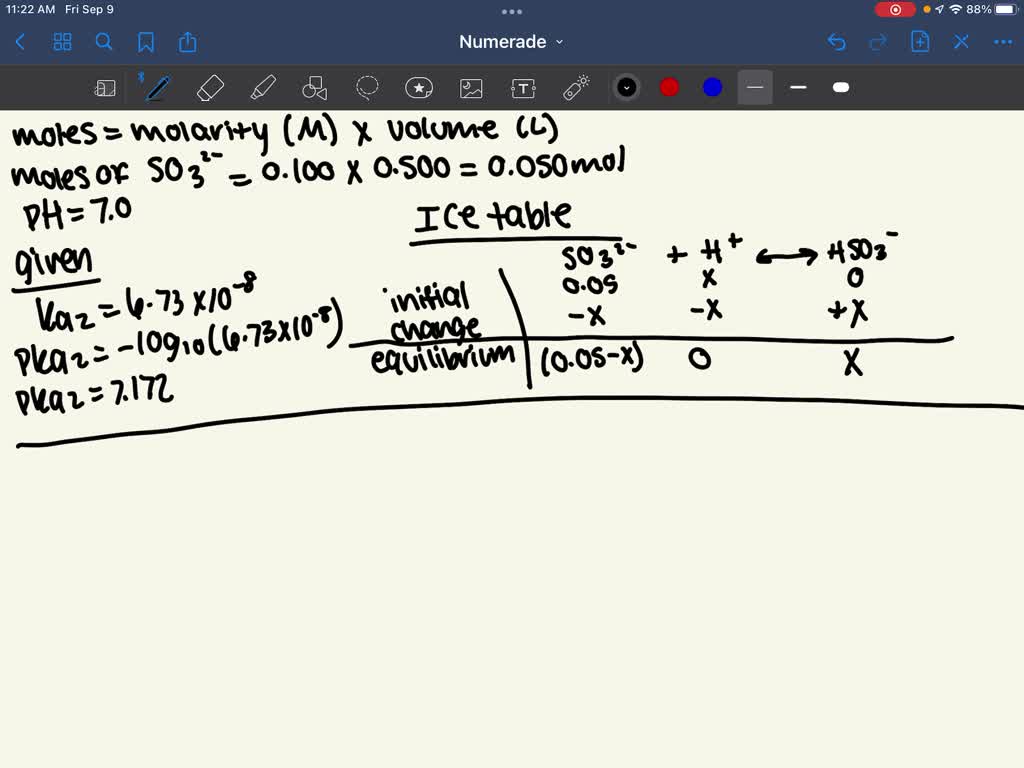
SOLVED: How many milliliters of 12.0 M HCl should be added to 5.00 x 102 mL of 0.100 M Na2SO3 to produce a pH 7.00 buffer solution. For H2SO3 Ka1 = 1.39 x 10-2 and Ka2 = 6.73 x 10-8.

Figure 10 from The initial phase of sodium sulfite pulping of softwood : A comparison of different pulping options | Semantic Scholar

Voltammograms measured in solutions of 0.05 М Na2SO3 + 0.1 M Na2SO4 at... | Download Scientific Diagram
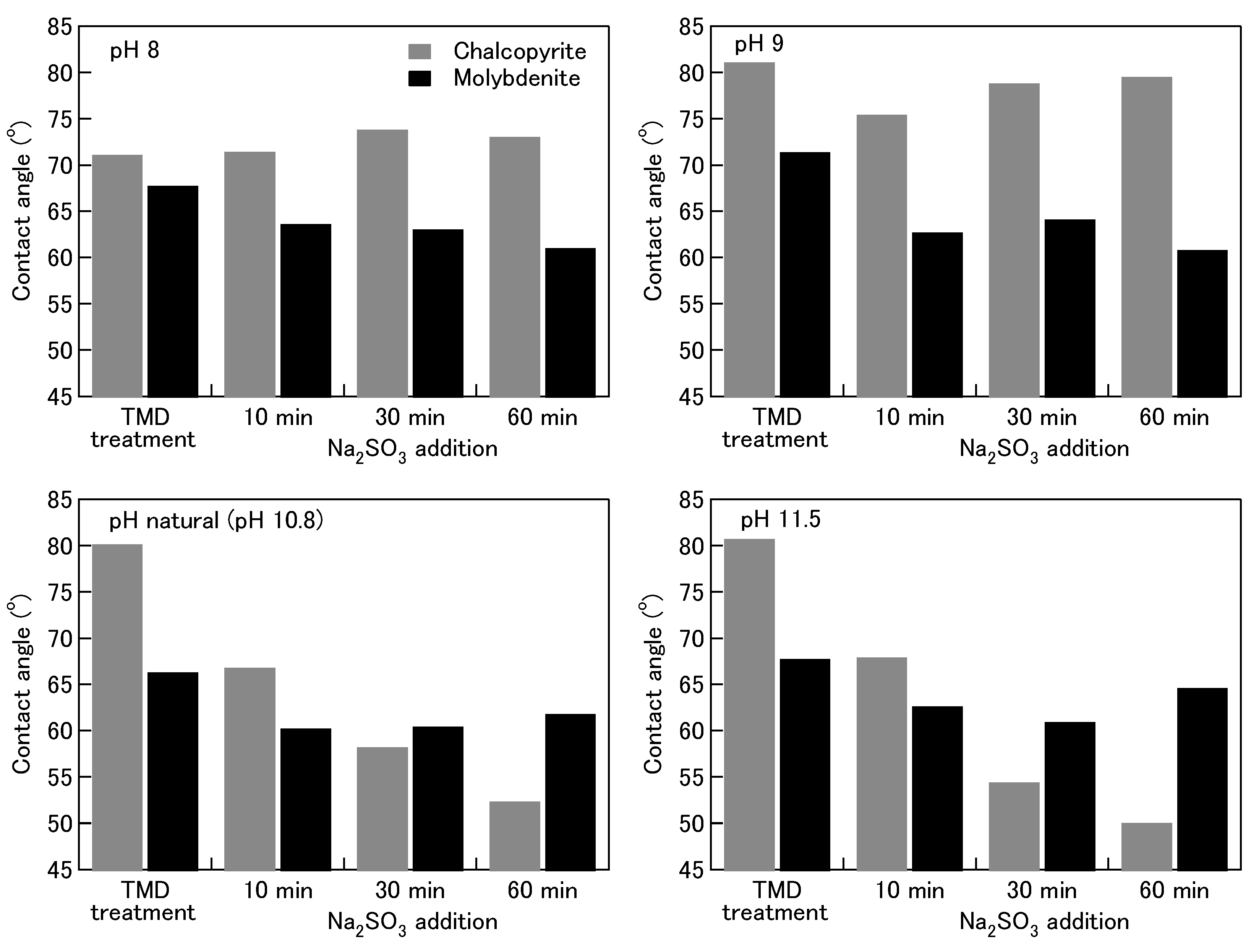
Minerals | Free Full-Text | Effect of Sodium Sulfite on Floatability of Chalcopyrite and Molybdenite