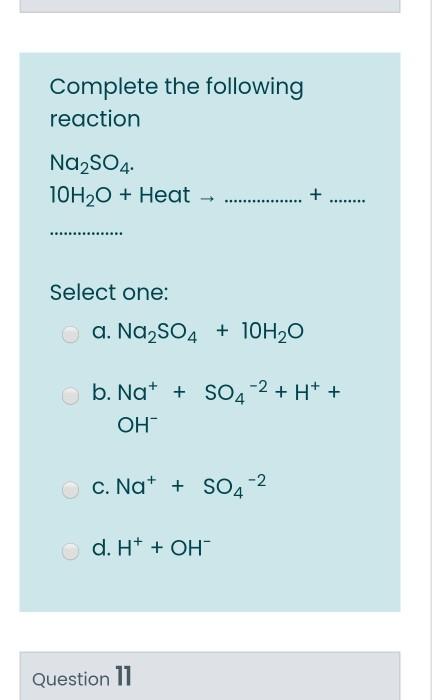
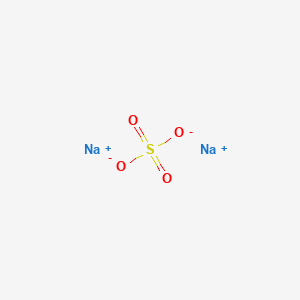
100 mL of a mixture of NaOH and Na2SO4 is neutralised by 10 mL of 0.5 M H2SO4 . Hence, NaOH in 100 mL solution is:

SOLVED: Sodium hydroxide (NaOl) reacts with sulfuric acid (IlzSO4) yields sodium sulfate (Na,SO4) and water. The unbalanced chemical equation is NaOH + HzS04 NazSO4 + Hz0 Balance the chemical equation using linear

Balance the following chemical equations:(a) HNO3 + Ca (OH)2→ Ca (NO3)2 + H2O (b) NaOH + H2SO4→Na2SO4 + H2O (c) NaCl + AgNO3→ AgCl + NaNO3 (d) BaCl2 + H2SO4→ BaSO4 + HCl

Reactions of Alkaline Minerals in the Atmosphere - Vargas Jentzsch - 2013 - Angewandte Chemie International Edition - Wiley Online Library

Sodium Sulfate (Na2SO4) - Structure, Meaning of Anhydrous, Properties, Preparations, Uses and FAQs of Sodium sulphate.
How many grams of sodium sulfate must be added to make 500 mL of a 0.25 M sodium sulfates solution? - Quora
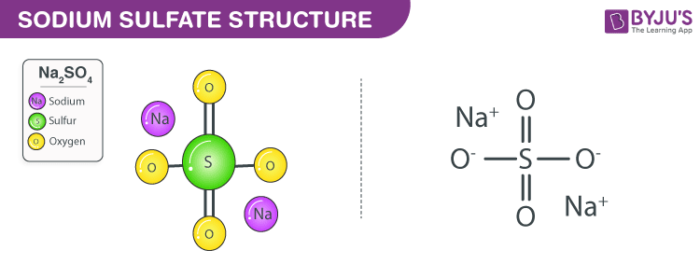
Sodium Sulfate (Na2SO4) - Structure, Meaning of Anhydrous, Properties, Preparations, Uses and FAQs of Sodium sulphate.
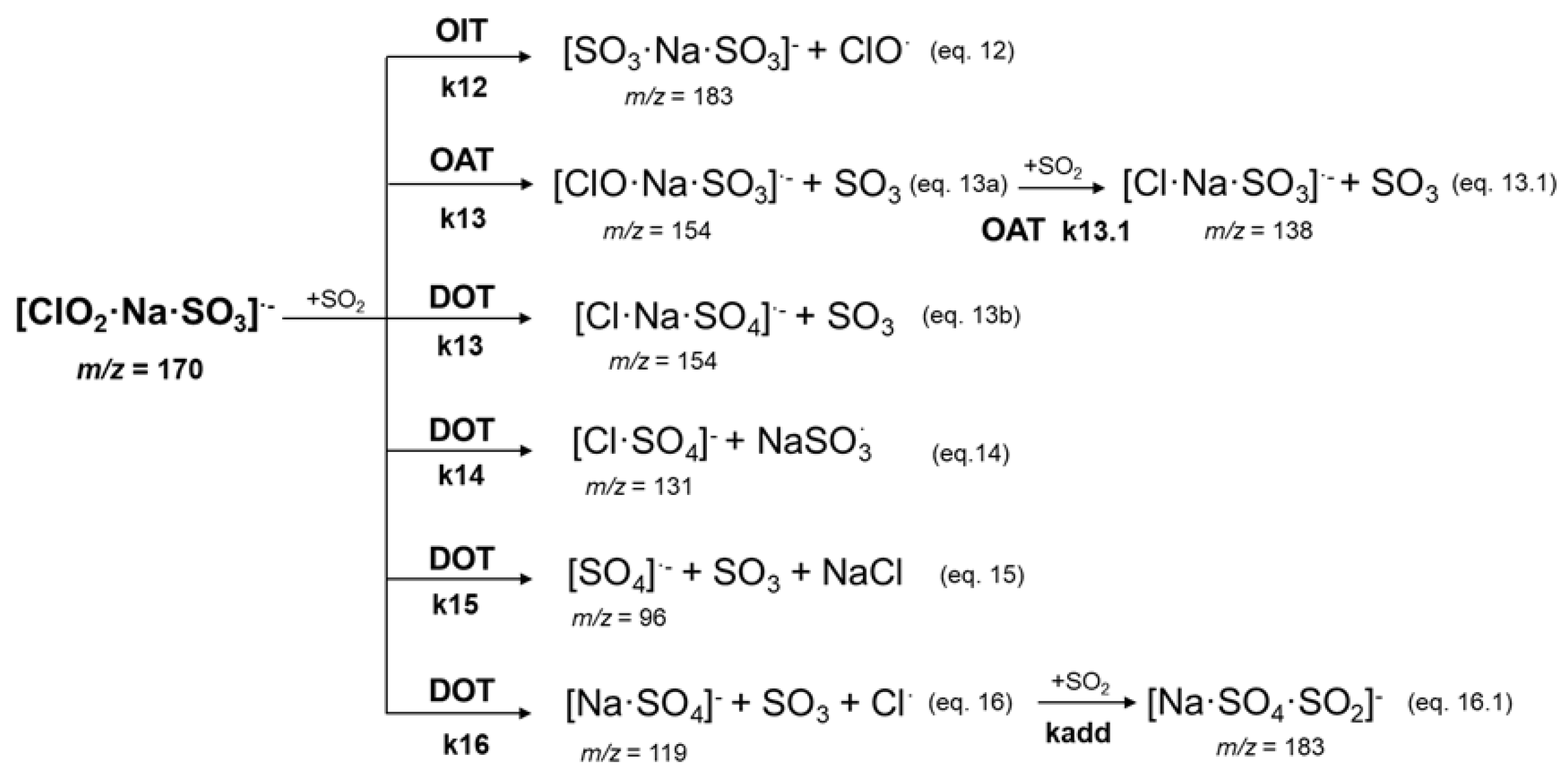
Molecules | Free Full-Text | Intracluster Sulphur Dioxide Oxidation by Sodium Chlorite Anions: A Mass Spectrometric Study

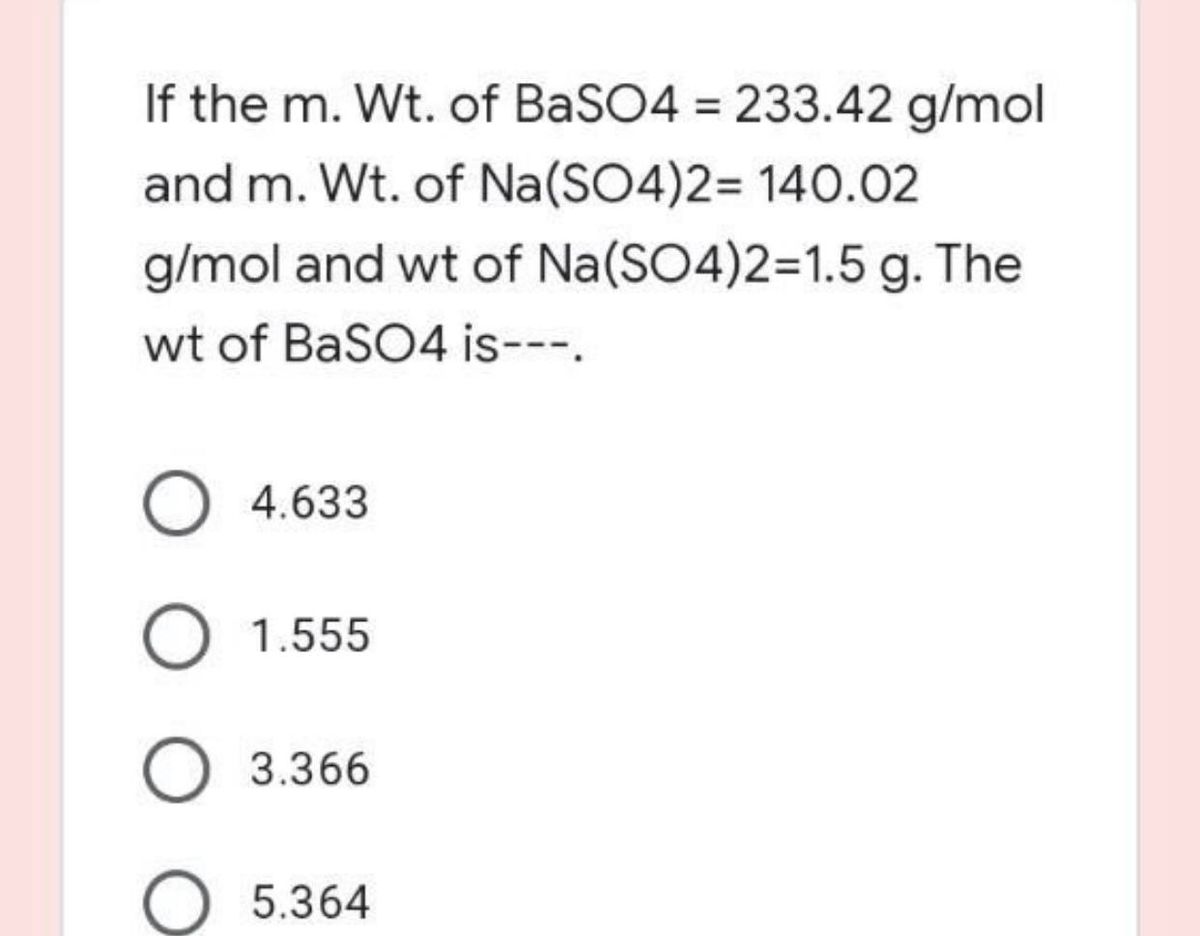




![ANSWERED] Be sure to answer all parts Write the net... - Physical Chemistry ANSWERED] Be sure to answer all parts Write the net... - Physical Chemistry](https://media.kunduz.com/media/sug-question-candidate/20220512084443228470-3529356.jpg)