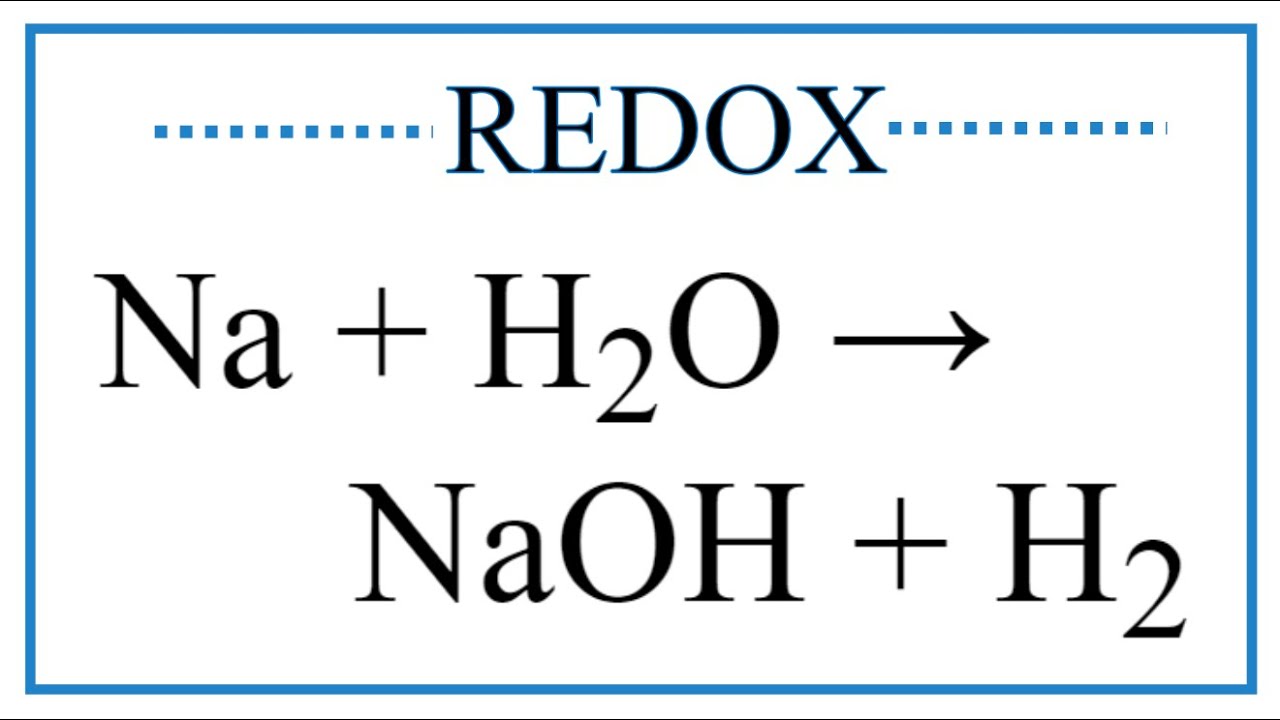
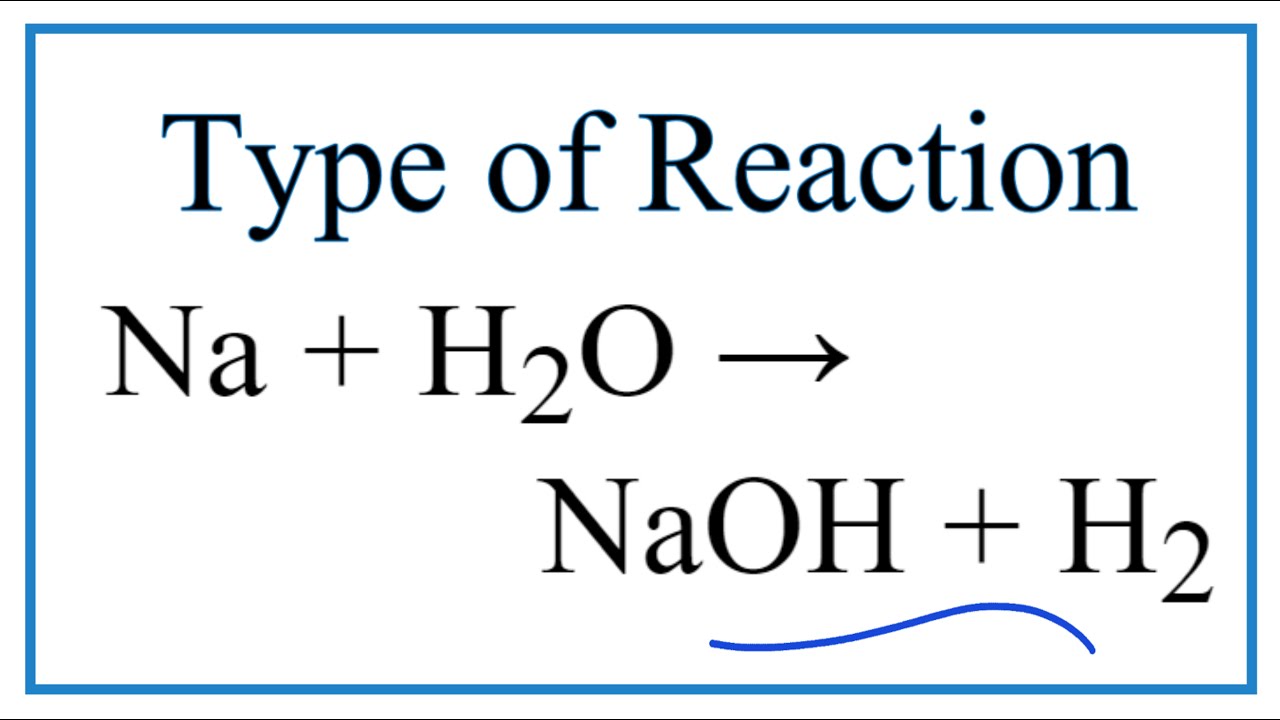
Write the balanced equation for the following chemical reactions:Sodium + Water → Sodium hydroxide + Hydrogen
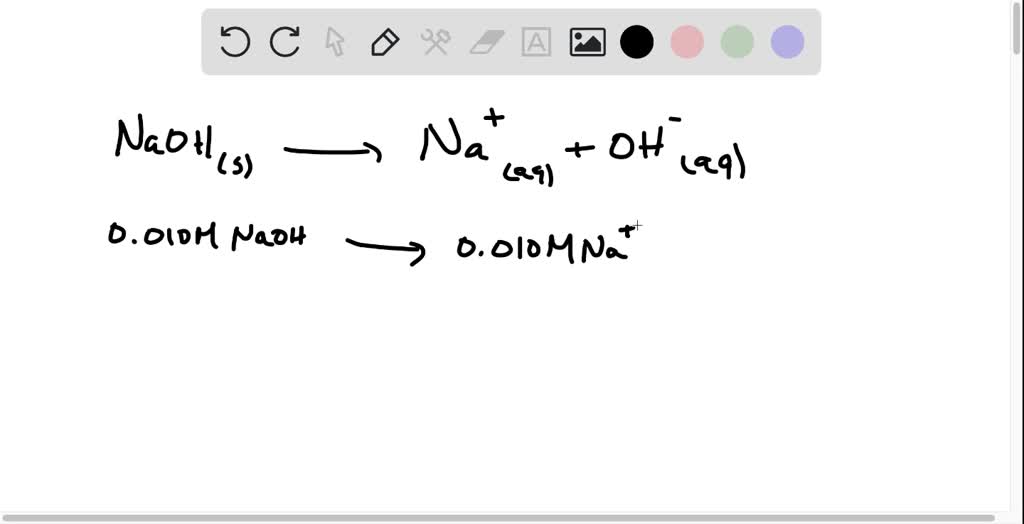
SOLVED: An aqueous solution of sodium hydroxide is labeled 0.010M NaOH. What is the molar concentration of NaOH, Na+, and OH- in the aqueous solution? Explain.
![How much of NaOH is required to neutralise 1500 cm³ of 0.1 N HCl (Na = 23) [KCET 2001] - Ranks Booster How much of NaOH is required to neutralise 1500 cm³ of 0.1 N HCl (Na = 23) [KCET 2001] - Ranks Booster](https://blogger.googleusercontent.com/img/b/R29vZ2xl/AVvXsEjJ7Doz4wL33X3oStv1SVBKLTGRXPwlDgkBIgJgFLvF75QitCj3uS2y9o9GJpO0kkC39QdennDvnw6NxVJGVSKai4Nur_fKeSUk_mpeII_n0iveg6xFV-aI5gegBQyGrMCGw34s_J4oKlwqiW1gJrmWScHkQPyFaUodnWagv9fllgFuIHSIhaCxbGtF/w0/How%20much%20of%20NaOH%20is%20required%20to%20neutralise%201500%20cm%C2%B3%20of%200.1%20N%20HCl%20(Na%20=%2023)%20%5BKCET%202001%5D.jpg)
How much of NaOH is required to neutralise 1500 cm³ of 0.1 N HCl (Na = 23) [KCET 2001] - Ranks Booster

Sodium orthosilicate, (mixture of NaOH and Na2SiO3 yielding Na4SiO4 in solution), Thermo Scientific Chemicals | Fisher Scientific

SOLVED: 0. Heory: #cz H,o + Naoh > Na (1h,02 Cefftrests . Azo VKaon #nl #tvahm MNe 0.0 ' M J l Naoh = M-V 46 H CzH,o = # | Naoh #
Calculating the mass of NaOH in 25 cm^3 of 0.2M sodium hydroxide solution(Na =23.0.O =16.0, H=1.0) - Tutorke