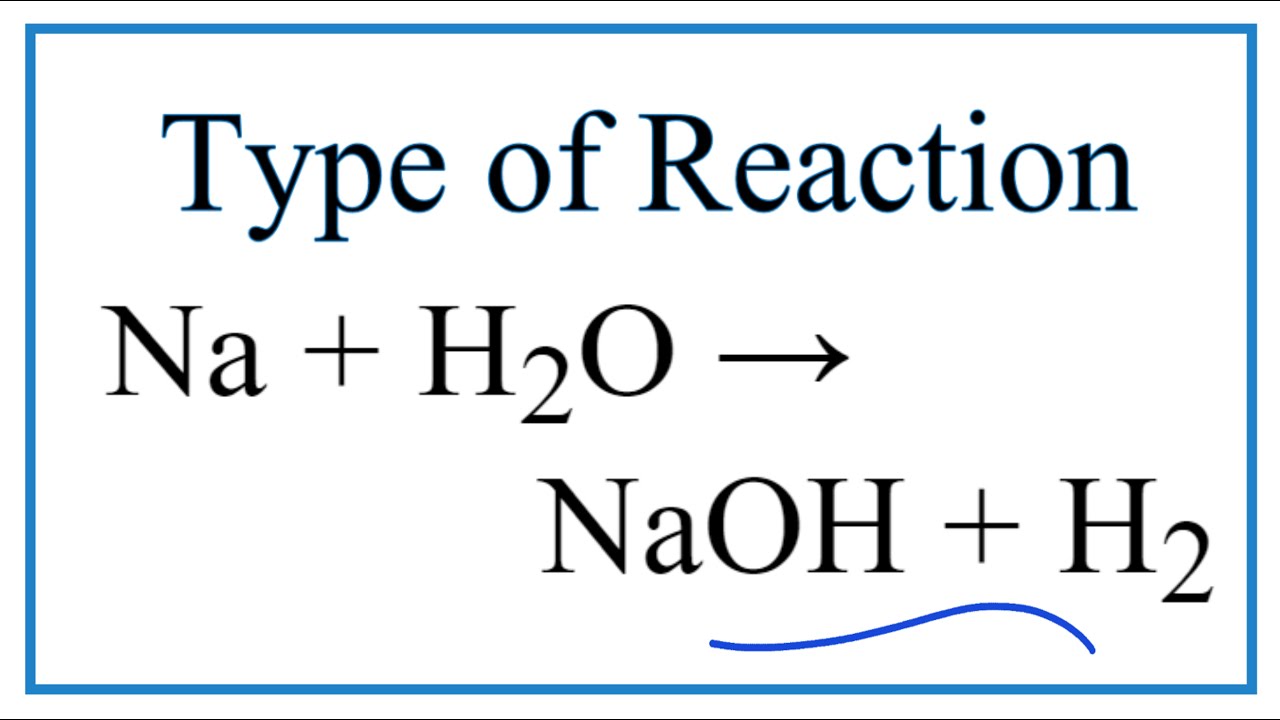
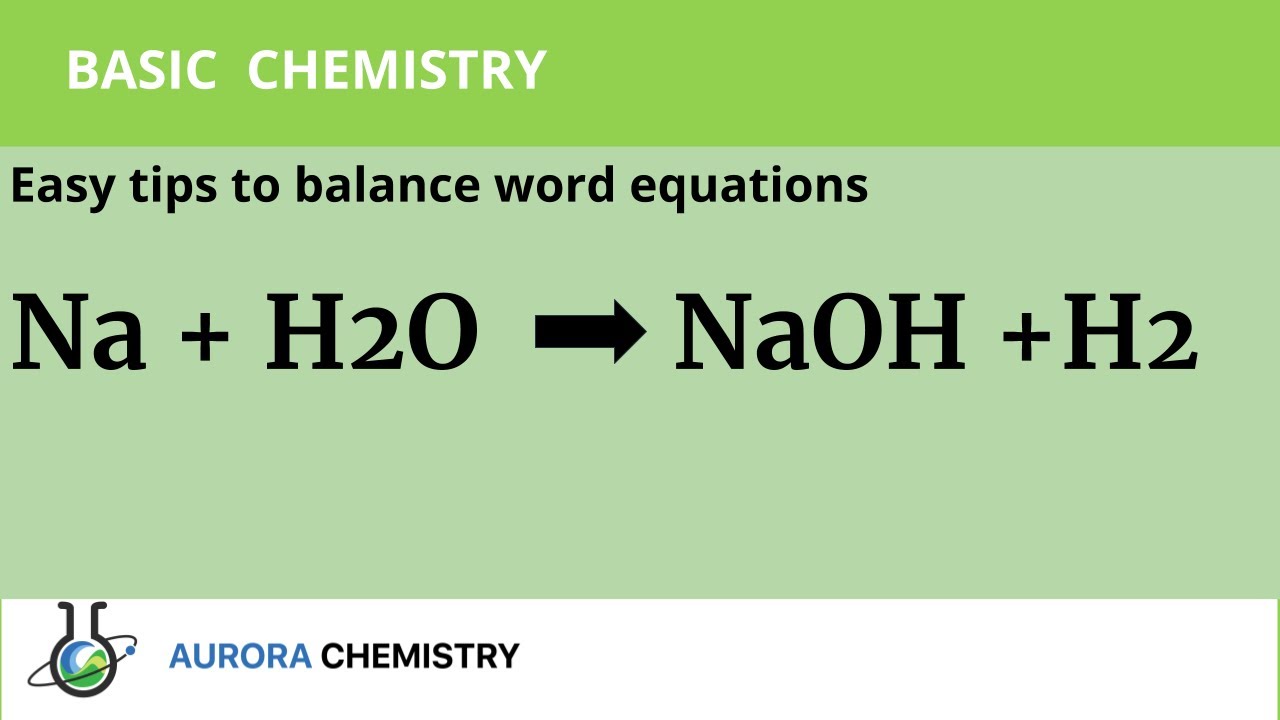
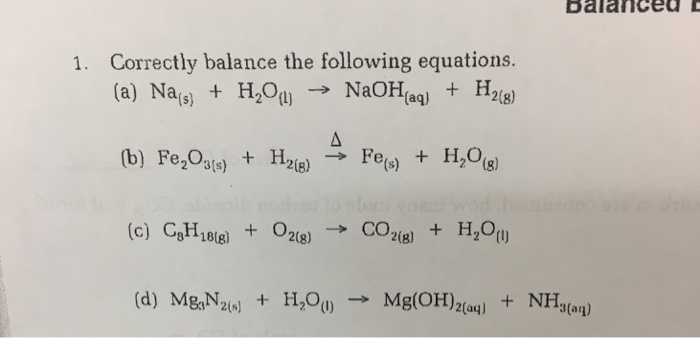Na +H20 --- NaOH + H2 (a) State the law that is followed by balancing a chemical equation (b) Balance - Science - Atoms and Molecules - 7937379 | Meritnation.com

10. Na + H2O ---> NAOH H2 11. H3PO4 + NaOH --- > Na3PO4 + H2O 12. CÓ + Fe3O4 CO2 + Fe .. 8,8 gam sắt(HI)sunfua у га. u huỳnh đã tham gia phản ứng. hiện tượ

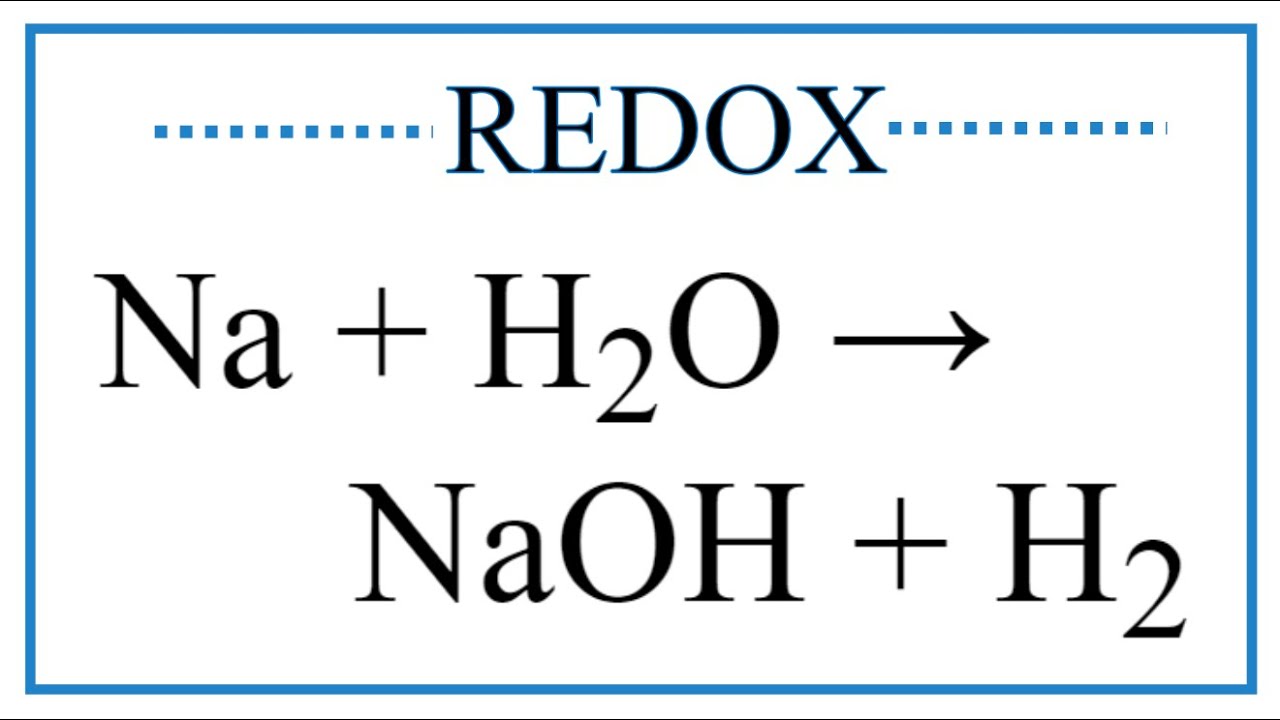
Расставьте коэффициенты в уравнении реакции используя метод электонного баланса - Na + H2O = NaOH - Школьные Знания.com
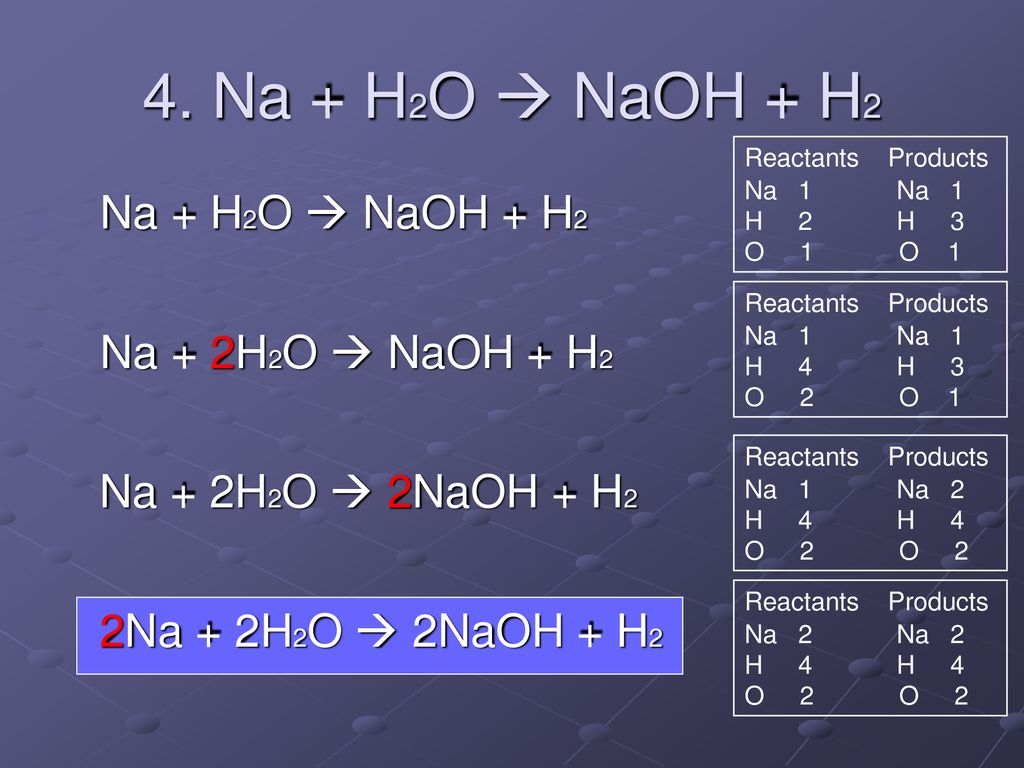
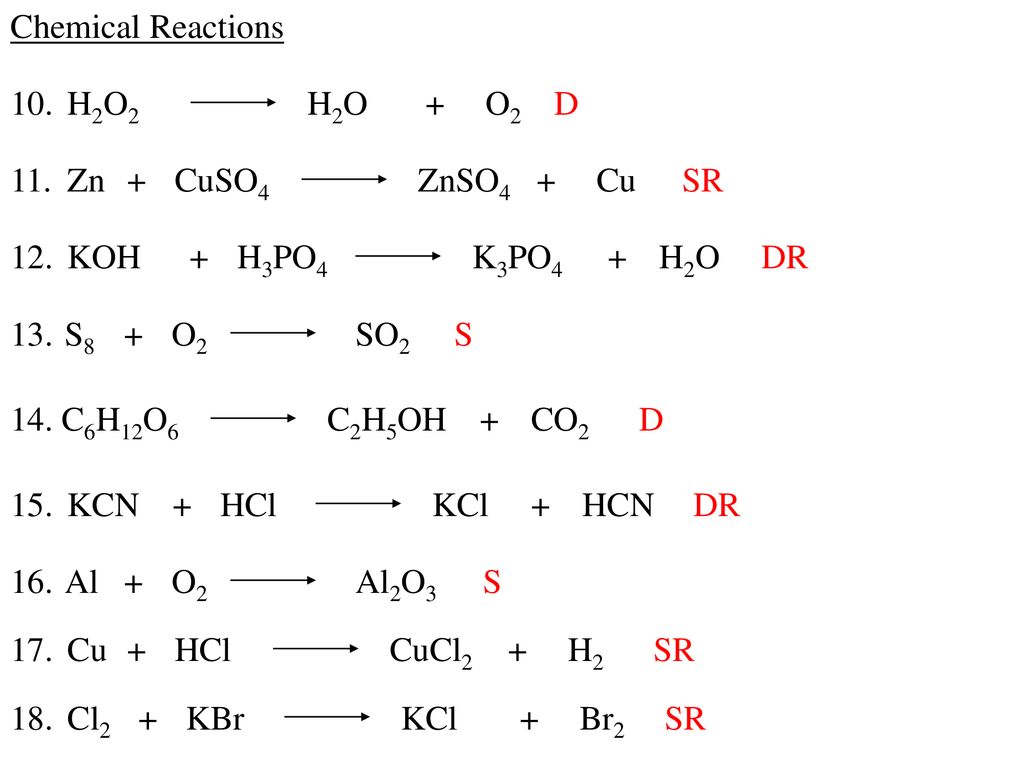
Chapter 6 – 3 Warm - Up 1. What are the 6 different types of chemical reactions we talked about in class? 2. What type of reaction is: 2 Na + Cl2 ppt download